Patient-Reported Outcome Measures to implement routine assessment of cancer survivors’ unmet needs: an overview of reviews and COSMIN analysis
Introduction
The number of cancer survivors (CSs) is increasing worldwide and is expected to reach 75 million by 2030 thanks to advances in treatments, screening techniques, and the ageing of the population.
Cancer care has traditionally focussed on diagnosis and life-saving treatments. However, research has shown that focusing on helping CSs cope with life beyond their acute treatment, i.e. cancer survivorship care, is just as important.
CSs experience a range of physical, psychosocial, spiritual, informational, and practical issues that may result in unmet needs, which are often neither identified nor addressed. Providing services relevant to their specific, unmet needs is crucial.
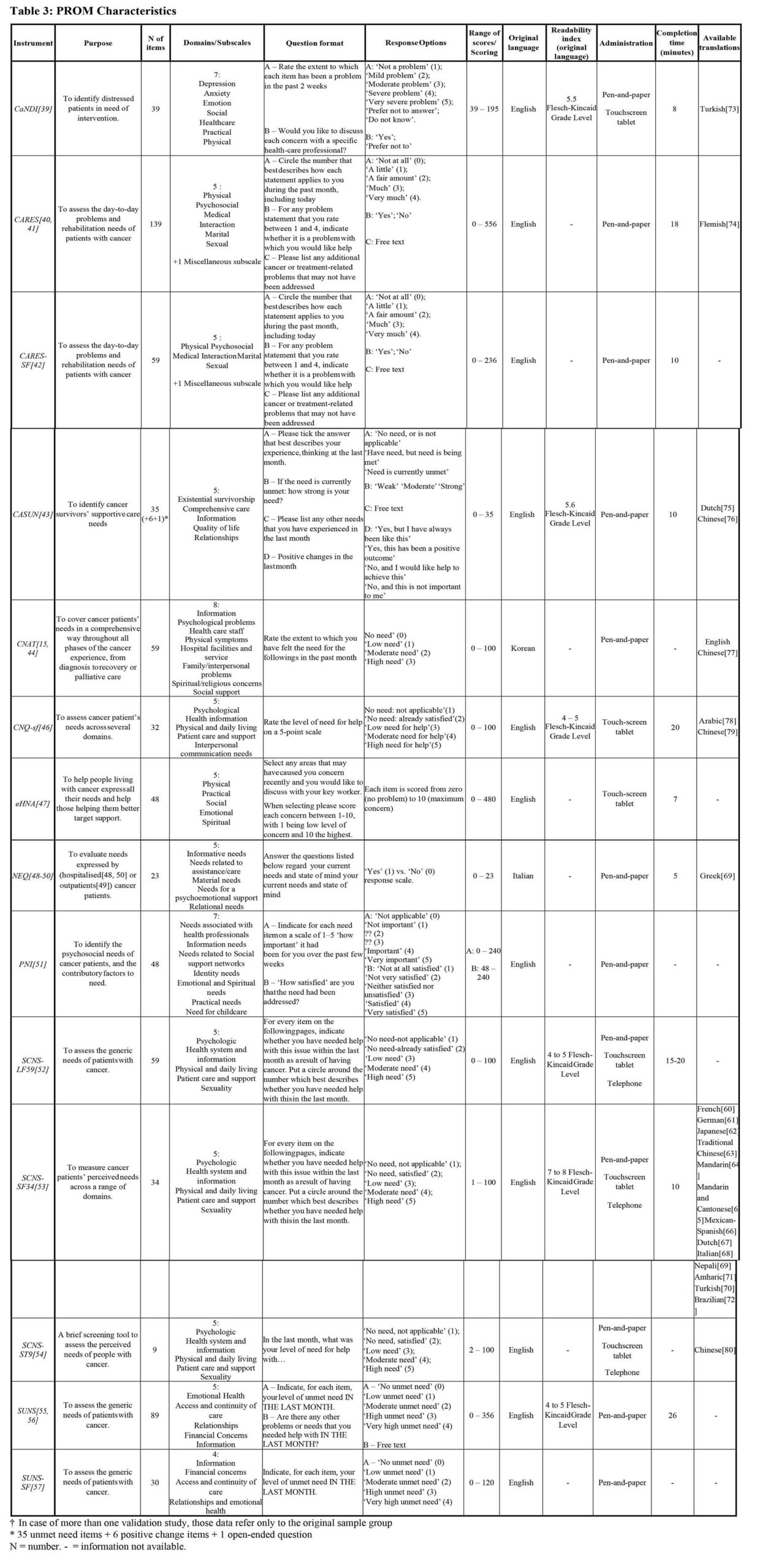
Patient-reported outcome measures (PROMs) are standardized, validated tools that give value to the information that comes from patients. There are currently various PROMs whose aim is to identify the unmet needs of CSs. Still, limited guidance supports choosing the most appropriate ones for this purpose.
Methods
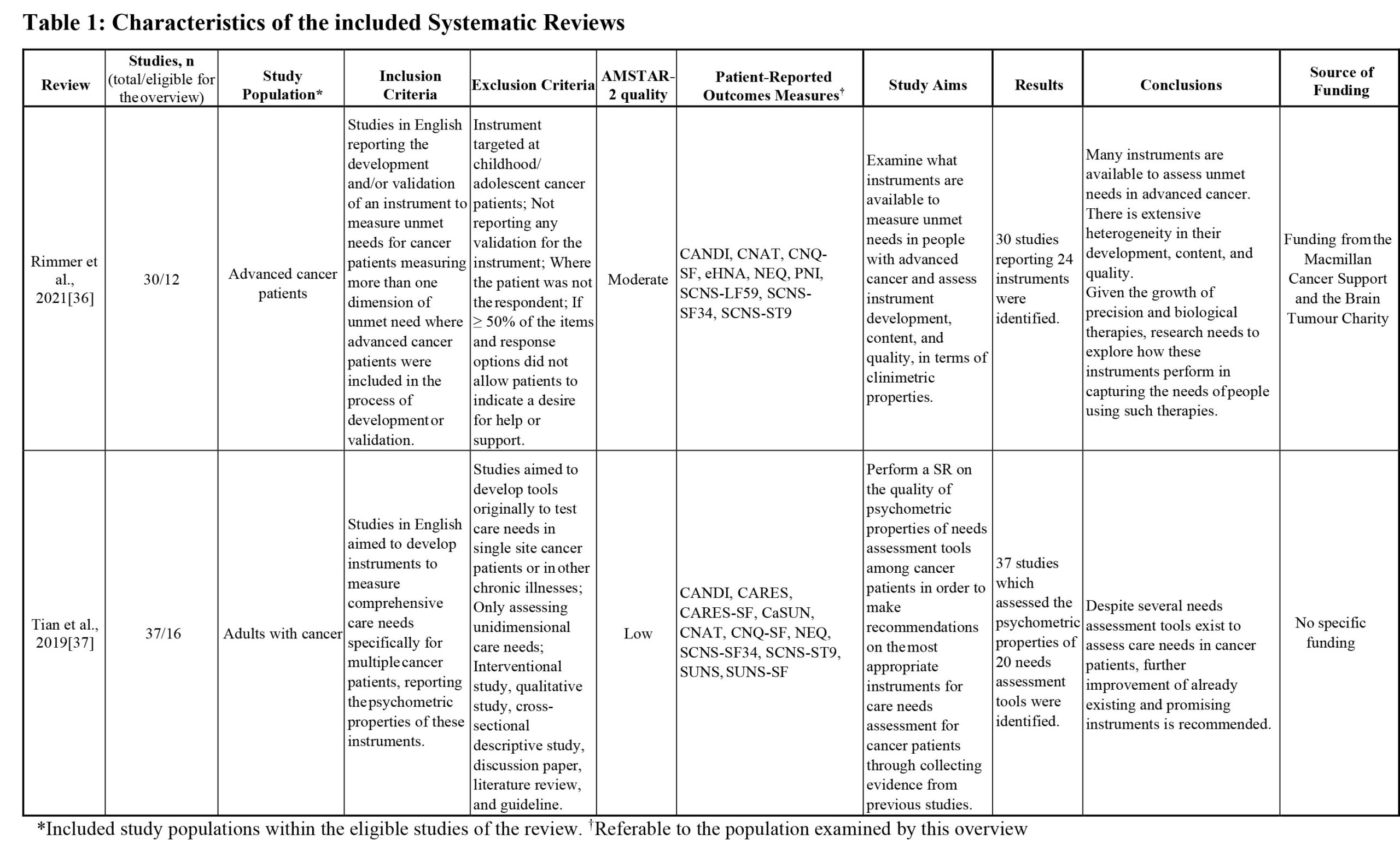
An Overview of Reviews was conducted to analyse systematic reviews (SRs) focusing on the psychometric properties of PROMs created to identify the unmet needs of adult CSs suffering from non-cutaneous cancer with a 5-year survival of ≥ 65% and an incidence of ≥ 5%.
A literature search was performed on the MEDLINE, EMBASE, Cochrane Database of Systematic Reviews, and CINAHL databases to identify SRs published between 2012 and January 2023.
The quality assessment of the included SRs was conducted by two independent reviewers using the AMSTAR-2 checklist and that of the primary studies using the COSMIN Risk of Bias checklist; the psychometric properties of all identified PROMs were evaluated according to the COSMIN updated quality criteria for good measurement properties.
These findings were then summarised, and their quality graded by means of the modified GRADE approach for grading the quality of the evidence in SRs of PROMs.
Results
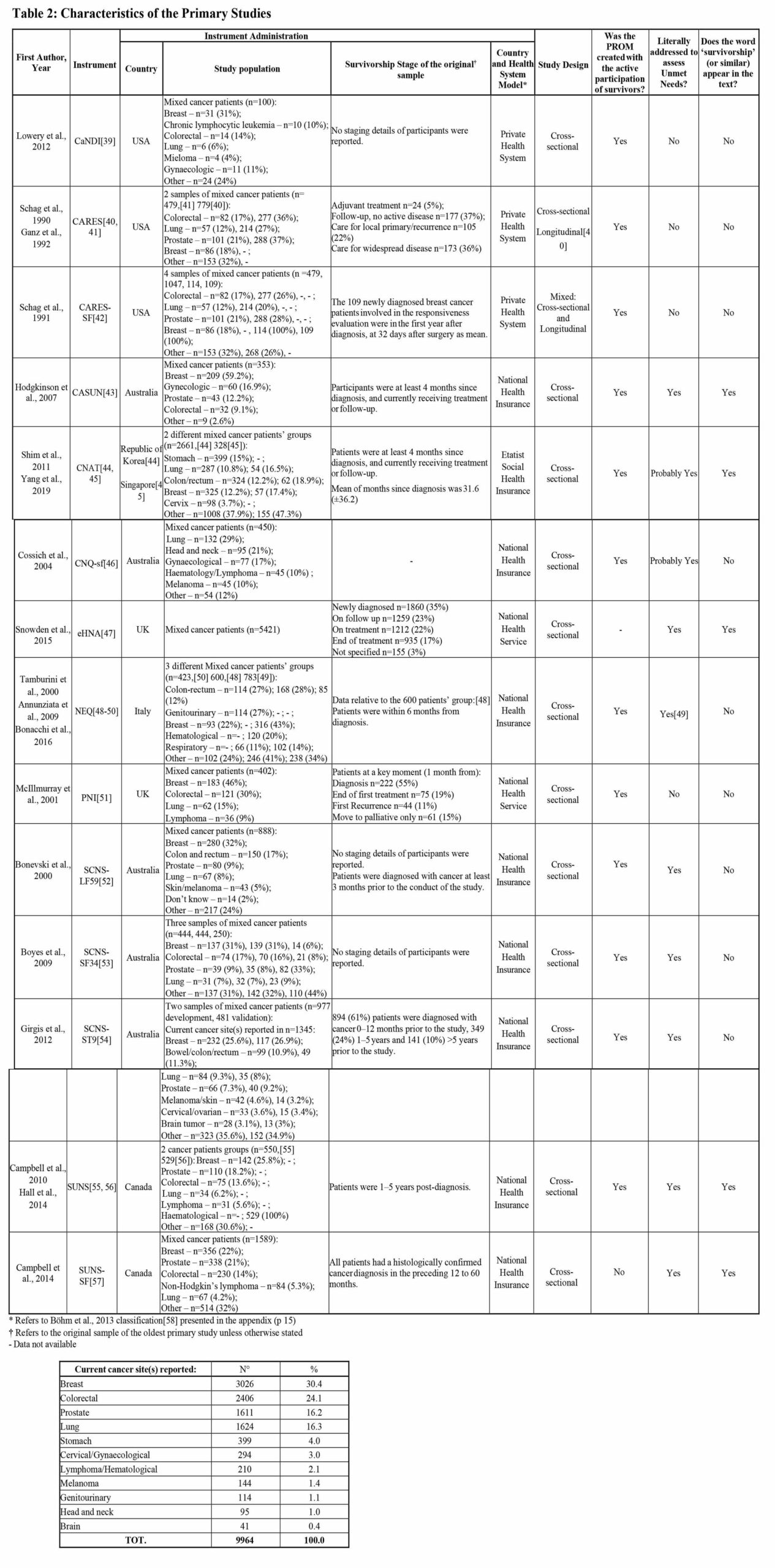
Two SRs, one of “moderate” and one of “low” quality according to the AMSTAR-2, were included. They covered 14 PROMs targeting our population of interest, described in 19 different articles, and tested on 19151 patients.
The methodological quality used to develop all the PROMs was rated as “inadequate”, primarily for not calculating measurement error (93% of the PROMs), since COSMIN Risk of Bias score applies the ‘worst score count’ rule.
Primary studies were inconsistent in reporting the psychometric properties of PROMs. However, those that did were judged positively, resulting “insufficient” in only 3% of the cases.
According to the GRADE approach, the level of evidence ranged from “very low” to “low”, with better grading for the Cancer Rehabilitation Evaluation System, followed by its short form (CARES-SF), and the Needs Evaluation Questionnaire.
Discussion and Conclusion
To exhaustively clarify the quality of psychometric properties of these PROMs, further studies are needed to investigate their incomplete aspects. However, the use of these PROMs in clinical practice and research is supported for those psychometric properties for which we have reliable reporting.
However, we recognize that the selection of the most appropriate PROMs to assess a domain of interest should be informed by the psychometric properties but should go beyond statistics, considering above all the context in which they will be applied.
Context refers not only to the pathology stage and environmental factors, but also to the geographical location, age, language, educational and socioeconomic level, and cultural background of the target population. The present descriptions of the contexts in which these tools have been developed and validated provides additional support in selecting specific PROMs based on content comparison.
REFERENCES
Rimmer B, Crowe L, Todd A, Sharp L. Assessing unmet needs in advanced cancer patients: a systematic review of the development, content, and quality of available instruments. J Cancer Surviv. 2021.
Tian L, Cao X, Feng X. Evaluation of psychometric properties of needs assessment tools in cancer patients: A systematic literature review. PLoS ONE. 2019;14.
Prinsen CAC, Mokkink LB, Bouter LM, Alonso J, Patrick DL, de Vet HCW, et al. COSMIN guideline for systematic reviews of patient-reported outcome measures. Quality of life research. 2018;27:1147-57.
Cieza A, Fayed N, Bickenbach J, Prodinger B. Refinements of the ICF Linking Rules to strengthen their potential for establishing comparability of health information. Disabil Rehabil. 2019;41:574-83.
Di Maio M, Basch E, Denis F, Fallowfield LJ, Ganz PA, Howell D, et al. The role of patient-reported outcome measures in the continuum of cancer clinical care: ESMO Clinical Practice Guideline. Ann Oncol. 2022;33:878-92.