ARE CLINICAL SENSORY TESTING (CST) SO ACCURATE TO EVALUATE SOMATOSENSORY SYSTEM DISORDERS? A SYSTEMATIC REVIEW
Autori
ROSSI FEDERICO [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
[Department of Biomedical and Neuromotor Sciences (DIBINEM), Alma Mater Studiorum, University of Bologna, Bologna, Italy]
CIOETA MATTEO [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
MONDELLI MARCO [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
ROSAFIO DANILO [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
CRICCO CHIARA [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
PELLICCIARI LEONARDO [IRCCS Istituto delle Scienze Neurologiche di Bologna, Bologna, Italy]
GIOVANNICO GIUSEPPE [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
BRINDISINO FABRIZIO [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
PRATO ILARIA [Department of Medicine and Health Science “Vincenzo Tiberio”, University of Molise, Campobasso, Italy]
Introduction to clinical case
The somatosensory system detects and interprets stimuli from both the external and internal environment through receptors, neural pathways, and cortical areas, enabling perception of touch, temperature, proprioception, and pain. Quantitative Sensory Testing (QST) is the gold standard for assessing somatosensory function and identifying dysfunctions such as hypoesthesia, allodynia, and hyperalgesia. Although effective, QST is costly and requires specialized training, limiting its routine clinical use. As an alternative, Clinical Sensory Testing (CST), or bedside sensory testing, has been developed as a more accessible and low-cost tool to evaluate sensory thresholds and predict treatment outcomes. This systematic review aims to assess the diagnostic accuracy of CST in identifying somatosensory disorders compared to standard laboratory methods.
Methods
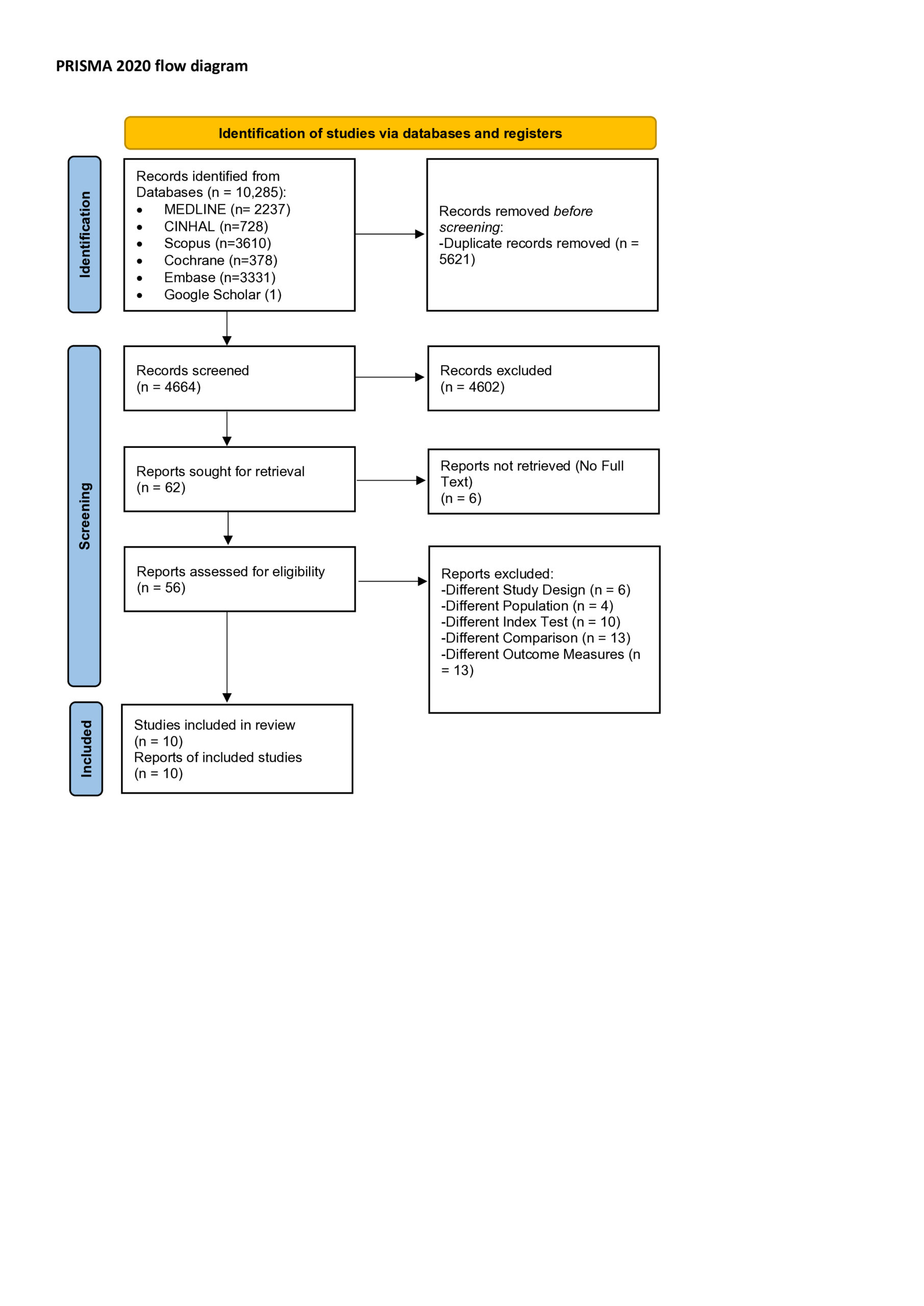
This systematic review will follow PRISMA-DTA guidelines and the Cochrane Handbook, including all relevant studies regardless of time, language, or location. Based on PICO criteria, it will evaluate the diagnostic accuracy (sensitivity and specificity) of Clinical Sensory Tests (CSTs) for somatosensory disorders, comparing them to other diagnostic tools like QST or skin biopsy. Bibliographic databases including Medline, CINHAL, PEdro, Web of Science, Cochrane Library, Scopus, Scholar and EMBASE will be consulted to investigate potentially relevant studies.The results of the bibliographic search will be uploaded to Rayyan. Two independent reviewers will handle study selection, data extraction, and risk of bias assessment, with a third reviewer resolving disagreements. Results will be synthesized narratively and in tables.
Results
Several Clinical Sensory Tests (CSTs) have shown varying levels of diagnostic accuracy in detecting somatosensory dysfunctions. The Semmes-Weinstein Monofilament Test and von Frey filaments demonstrated high specificity, especially when compared to gold standards like Nerve Conduction Studies and MRI. Tools like the Neuropen, tuning forks, Q-tip, and temperature-based tests also showed promising sensitivity or specificity depending on the context. However, many CSTs showed a trade-off between sensitivity and specificity, and outcomes varied based on methodology and condition assessed.
Discussion and clinical relevance
Identifying somatosensory dysfunctions is essential for improving diagnosis and treatment. Many authors stress the importance of developing fast, low-cost tests that can assess all nerve fiber types without requiring complex equipment. Tests like the SWMT and StEP showed good sensitivity and specificity, but other tools yielded inconsistent results. However, population heterogeneity and lack of standardization in test administration currently limit CSTs from being considered fully reliable diagnostic methods.
REFERENCES
- Zhu GC, Böttger K, Slater H, Cook C, Farrell SF, Hailey L, Tampin B, Schmid AB. Concurrent validity of a low-cost and time-efficient clinical sensory test battery to evaluate somatosensory dysfunction. Eur J Pain. 2019 Nov;23(10):1826-1838.
- Scholz J, Mannion RJ, Hord DE, Griffin RS, Rawal B, Zheng H, Scoffings D, Phillips A, Guo J, Laing RJ, Abdi S, Decosterd I, Woolf CJ. A novel tool for the assessment of pain: validation in low back pain. PLoS Med. 2009 Apr 7;6(4):e1000047. doi: 10.1371/journal.pmed.1000047. Epub 2009 Apr 7. PMID: 19360087; PMCID: PMC2661253.
- Aithal, V., Bhat, S. Semmes Weinstein monofilament test for detection of diabetic peripheral neuropathy: sensitivity and specificity. Egypt J Neurol Psychiatry Neurosurg60, 6 (2024).
- Ridehalgh C, Sandy-Hindmarch OP, Schmid AB. Validity of Clinical Small-Fiber Sensory Testing to Detect Small-Nerve Fiber Degeneration. J Orthop Sports Phys Ther. 2018 Oct;48(10):767-774. doi: 10.2519/jospt.2018.8230. Epub 2018 Jun 22. PMID: 29932873.
- Lin, JH., Hsieh, YC., Chen, YC. et al.Diagnostic accuracy of standardised qualitative sensory test in the detection of lumbar lateral stenosis involving the L5 nerve root. Sci Rep 7, 10598 (2017).